Get Ready For A New Innovation In Lashes.

By Milad Estivan
CEO/Member of Pan Pacific Laser Vision Center
The only FDA approved eyelash product. Prescription only and physician administered.
LATISSETM (bimatoprost ophthalmic solution) 0.03% is the first and only prescription treatment approved by the FDA for inadequate or not having enough eyelashes, growing them longer, fuller and darker. Not like other products LATISSETM works effectively and safety.
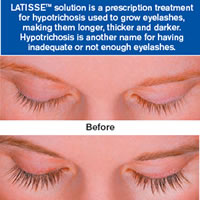
LATISSETM makes lash growth possible because of its active ingredient: bimatoprost. Although the precise mechanism of action is not known, research suggests that the growth of eyelashes occurs by increasing the percent of hairs in, and the duration of, the anagen (or growth) phase. Lashes can grow longer, thicker and darker because bimatoprost can also prolong this growth phase. Clinically proven results you can see over time.
LATISSETM is easy to use, FDA approved and effective. It’s a once-a-day treatment you apply topically to the base of your upper eyelashes. Patients in a clinical trial saw results gradually over time (starting at 4 weeks with full results after 16 weeks). It’s an innovation backed by research.

While LATISSETM is a new treatment in eyelash growth, it was developed through years of research by Allergan, a pharmaceutical leader with over 60 years of expertise in prescription eye care products.
To find out if Latisse solution is right for you, you will undergo a simple eye exam conducted by our board certified ophthalmologist Dr. Stephen Gee, MD. Dr. Gee will meet with you at 2 months and 4 months after you start Latisse to assure safety and effectiveness of the product.
For a free consultation call us at 949-9200 or visit our website at www.panpacificlaser.com.
E-mail this story | Print this page |
Comments (0) | Archive |  | RSS
| RSS
Most Recent Comment(s):




 Del.icio.us
Del.icio.us